Zymolyase® from Arthrobacter luteus
 Zymolyase®, produced by a submerged culture of Arthrobacter luteus(1), has strong lytic activity against living yeast cell walls(2),(3) to produce protoplast or spheroplast of various strains of yeast cells. An essential enzyme for the lytic activity of Zymolyase® is beta.-1,3-glucan laminaripentaohydrolase. It hydrolyzes linear glucose polymers at beta.-1,3-linkages and releases specifically laminaripentaose as the main and minimum product unit(4), (5), (10), (11).
Zymolyase®, produced by a submerged culture of Arthrobacter luteus(1), has strong lytic activity against living yeast cell walls(2),(3) to produce protoplast or spheroplast of various strains of yeast cells. An essential enzyme for the lytic activity of Zymolyase® is beta.-1,3-glucan laminaripentaohydrolase. It hydrolyzes linear glucose polymers at beta.-1,3-linkages and releases specifically laminaripentaose as the main and minimum product unit(4), (5), (10), (11).
There are two preparations of Zymolyase®, Zymolyase®-20T and Zymolyase®-100T, having lytic activity of 20,000 units/g and 100,000 units/g respectively. Zymolyase®-20T is ammonium sulfate precipitate while Zymolyase®-100T is a further purified preparation by affinity chromatography(9). Lytic activity varies depending on yeast strain, growth stage of yeast, or cultural conditions(6-8). Further information related to Zymolyase® can be obtained in the reference section below(12-16).
Electron microscope images of yeast cell
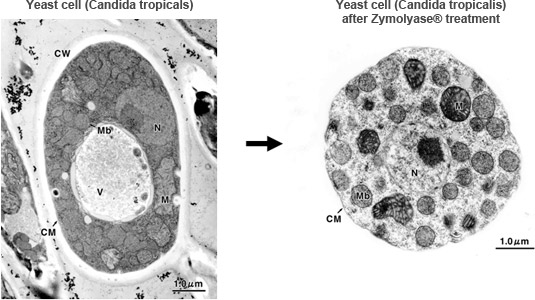
CW: Cell Wall CM: Cell Membrane M: Mitochondria Mb: Microbody N: Nucleus V: Vacuole
Data courtesy of Professor Masako Osumi, Nippon Women's University
Product Information
| Zymolyase®-20T | Zymolyase®-100T | ||
|---|---|---|---|
| Form | Lyophilized Powder | ||
| Purification | Ammonium Sulfate Precipitation | Affinity Chromatography | |
| Activity | 20,000 units/g | 100,000 units/g | |
| Essential enzyme | Beta-1,3-glucan laminaripentaohydrolase | ||
| Other activities contained *1 | Beta-1,3-glucanase | ca. 1.5 x 106 units/g | ca. 1.0 x 107 units/g |
| Protease | ca. 1.0 x 104 units/g | ca. 1.7 x 104 units/g | |
| Mannanase | ca. 1.0 x 106 units/g | ca. 6.0 x 104 units/g | |
| Contaminants | Amylase, Xylanase, Phosphatase | Trace amount | Non detectable |
| Optimum pH & Temperature | pH 7.5, 35°C (for lysis of viable yeast cells) pH 6.5, 45°C (for hydrolysis of yeast glucan) |
||
| Stability | 4°C | No loss of activity was found after storage for 1 year | |
| Heat stability | 30°C | 70% of lytic activity is lost after storage for 3 months | 90% of lytic activity is lost after storage for 3 months |
| 60°C | Lytic activity is lost on incubation for 5 minutes | ||
| Specificity (Lytic Spectrum) | Ashbya, Candida, Debaryomyces, Eremothecium, Endomyces, Hansenula, Hanseniaspora, Kloeckera, kluyveromyces, Lipomyces, Metschnikowia, Pichia, Pullularia, Torulopsis, Saccharomyces, Saccharomycopsis, Saccharomycodes, Schwanniomyces, etc. | ||
| Activators | Cystein, 2-mercaptoethanol, Dithiothreitol etc. | ||
*1 See reference, Kitamura, K., Kaneko, T. and Yamamoto, Y.: J. Gen. Appl. Microbiol., 18, 57 (1972) as to the definition of each enzyme units.
Precaution on use
- Avoid using nitrocellulose filters and use of material other than nitrocellulose when sterilizing. Zymolyase® may be adsorbed on nitrocellulose membranes.
- Zymolyase® -100T may not be completely dissolved in buffers. Use Zymolyase® as suspension.
- When sterilized, Zymolyase® is used in a concentration higher than 0.05%, prepare 2% Zymolyase® solution in buffers containing 5% glucose, filter the suspension and dilute the solution with the appropriate buffer.
Unit Definition
One unit of lytic activity is defined as that amount which indicates 30% of decrease in absorbance at 800 nm (A800) of the reaction mixture under the following condition
Reaction mixture
| Enzyme solution | 1 ml (0.05-0.1 mg/ml for Zymolyase®-20T) 1 ml (0.012-0.024 mg/ml for Zymolyase®-100T) |
|---|---|
| Brewer's yeast cell suspension | 3 ml (2 mg/ml) |
| M/15 Phosphate buffer, pH 7.5 | 5 ml |
| Distilled water | 1 ml |
After incubation for 2 hours at 25°C with gentle shaking, A800 of the mixture is determined. When 60% of A800 decrease, equivalent to 2 units, is observed in the reaction system, the brewer's yeast cells are completely lysed, namely 1 unit of Zymolyase® lyses 3 mg dry weight of brewer's yeast.
References
- Cobos, Samantha N., et al. "[PRION+] States Are Associated with Specific Histone H3 Post-Translational Modification Changes." Pathogens 11.12 (2022): 1436.
- Kaur, Hardeep, G. N. Krishnaprasad, and Michael Lichten. "A RAD9-dependent cell cycle arrest in response to unresolved recombination intermediates in Saccharomyces cerevisiae." bioRxiv (2019): 736850.
-
Kaneko, T., Kitamura, K and Yamamoto, Y.: J. Gen. Appl. Microbiol., 15, 317 (1969)
-
Kitamura, K., Kaneko, T. and Yamamoto, Y.: Arch. Biochem. Biophys., 145, 402 (1971)
-
Kitamura, K., Kaneko, T. and Yamamoto, Y.: J. Gen. Appl. Microbiol., 18, 57 (1972)
-
Kitamura, K. and Yamamoto, Y.: Arch. Biochem. Biophys., 153, 403 (1972)
-
Kaneko, T., Kitamura, K. and Yamamoto, Y.: Agric. Biol. Chem., 37, 2295 (1973)
-
Kitamura, K., Kaneko, T. and Yamamoto, Y.: J. Gen Appl. Microbiol., 20, 323 (1974)
-
Kitamura, K. and Yamamoto,.: Agric. Biol. Chem., 45, 1761 (1981)
-
Katamura, K. and Tanabe, K.: Agric, Biol. Chem., 46, 553 (1982)
-
Katamura, K.: J. Ferment. Technol., 60, 257 (1982)
-
Kitamura, K.: Agric. Biol. Chem., 46, 963 (1982)
-
Kitamura, K.: Agric. Biol. Chem., 46, 2093 (1982)
-
Calza R. E., Schroeder A. L.: J. Gen. Microbiol., 129, 413 (1983)
-
Iizuka Masaru, Torii Yasuhiko, Yamamoto Takehiko: Agric. Biol. Chem., 47 (12), 2267 (1983)
-
Shibata Nobuyuki, Kobayashi Hidemitsu, tojo Menehiro, Suzuki Shigeo: Arch. Biochem. Biophys., 251(2), 697 (1986)
-
Iijima Y., Yanagi S. O.: Agric. Biol. Chem., 50 (7), 1855 (1986)
-
Herrero Enrique, Sanz Pascual. Sentandreu Rafael: J. Gen. Microbiol., 133 (10), 2895 (1987)
