Column Selection Guide
Reversed Phase Specialty Columns
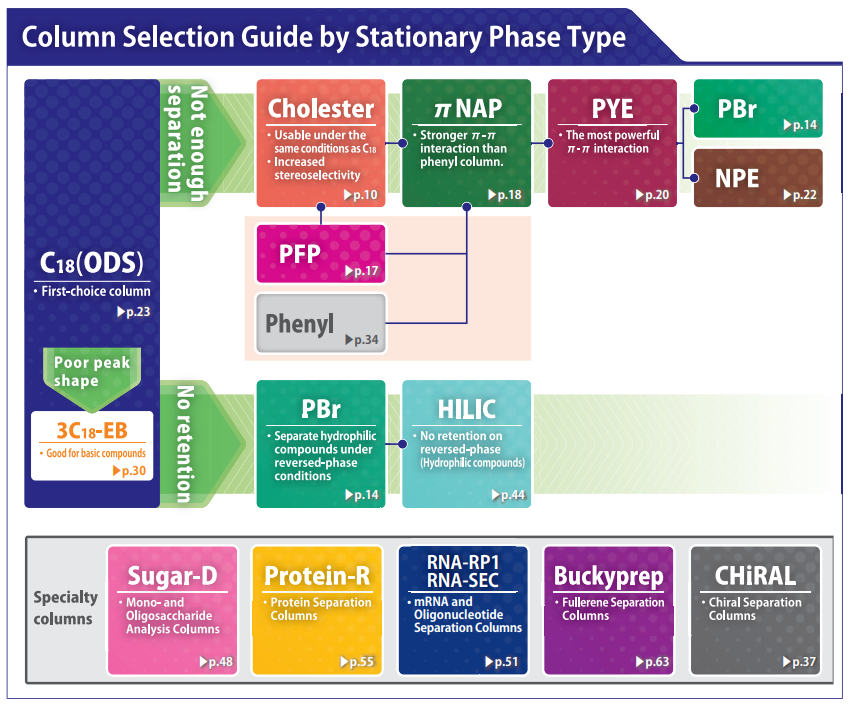
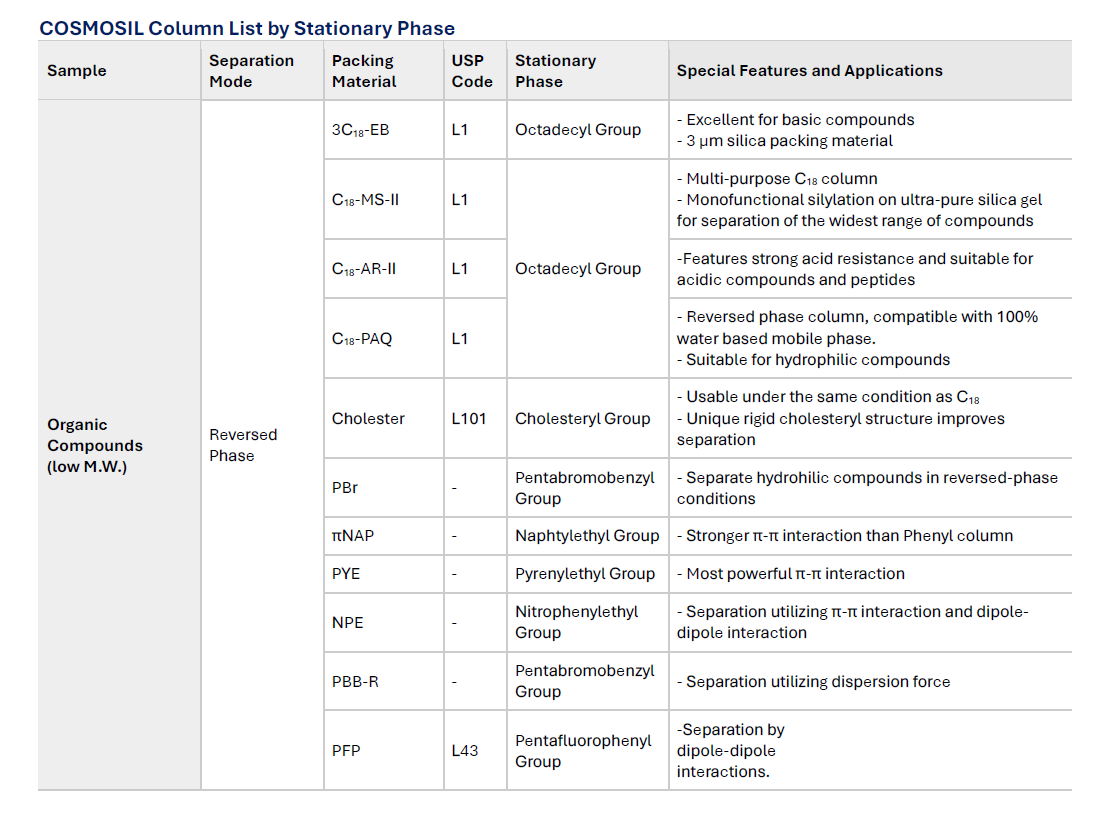
Reversed phase HPLC columns have been widely used because of their superior resolution, high theoretical plate number and ease of use. Since hydrophobic interaction is the dominant separation mechanism in reversed phase chromatography, conventional stationary phases such as C18 and C18 do not offer optimum selectivity for compounds with similar hydrophobicity. COSMOSIL offers a broad selection of columns with unique stationary phases for separation of these difficult analytes. These columns offer improved separation of structurally similar compounds that are difficult to analyze with a C18 type column.
Specifications
| Packing Material | Cholester | PBr | PFP | πNAP | PYE | NPE | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Silica Gel | Fully-Porous | Core-Shell | Fully-Porous | Core-Shell | Fully-Porous | Fully-Porous | Fully-Porous | Fully-Porous | ||
| Particle Size | 2.5 µm | 5 µm | 2.6 µm | 5 µm | 2.6 µm | 5 µm | 2.5 µm | 5 µm | 5 µm | 5 µm |
| Pore Size | 130Å | 120Å | 90Å | 120Å | 90Å | 120Å | 130Å | 120Å | 120Å | 120Å |
| Specific Surface Area | 300 m2/g | 300 m2/g | 150 m2/g | 300 m2/g | 150 m2/g | 300 m2/g | 300 m2/g | 300 m2/g | 300 m2/g | 300 m2/g |
| Stationary Phase | Cholesteryl Group | Pentabromobenzyl group | Pentafluorophrnyl group | Naphthylethyl Group |
Pyrenylethyl Group |
Nitrophenylethyl Group |
||||
| Bonding Type | Monomeric | |||||||||
| Main Interaction |
- Hydrophobic Interaction
- Molecular Shape Selectivity
|
- Hydrophobic interaction
- Dispersion force
|
- Hydrophobic interaction
- π-π Interaction
- Dipole-dipole Interaction
|
- Hydrophobic Interaction
- π-π Interaction
|
- Hydrophobic Interaction
- π-π Interaction
- Dispersion Force
- Molecular shape selectivity
|
- Hydrophobic Interaction
- π-π Interaction
- Dipole-dipole Interaction
|
||||
| End Capping Treatment | Near-perfect treatment | |||||||||
| Carbon Content | 21% | 20% | - | 8% | - | 10% | 14% | 11% | 18% | 9% |
| pH Range | 2–7.5 | |||||||||
| Feature |
- Usable under the same conditions as C18
- High molecular shape selectivity
|
- Separate hydrophilic compounds in reversed phase conditions
- Separate using dispersion force
|
- Weak dipole-dipole interaction
|
- Stronger π-π interaction than phenyl column
|
- Very strong π-π interaction
|
- Strong dipole-dipole interaction
|
||||
Fully-Porous: High purity porous spherical silica / Core-Shell: Core-Shell silica gel
Selectivity for positional isomers of dinitrobenzene
Different stationary phase exhibits different selectivity due to the use of forces that C18 (hydrophobic interaction) does not have. By using these columns, you can achieve separation that cannot be done using only C18.
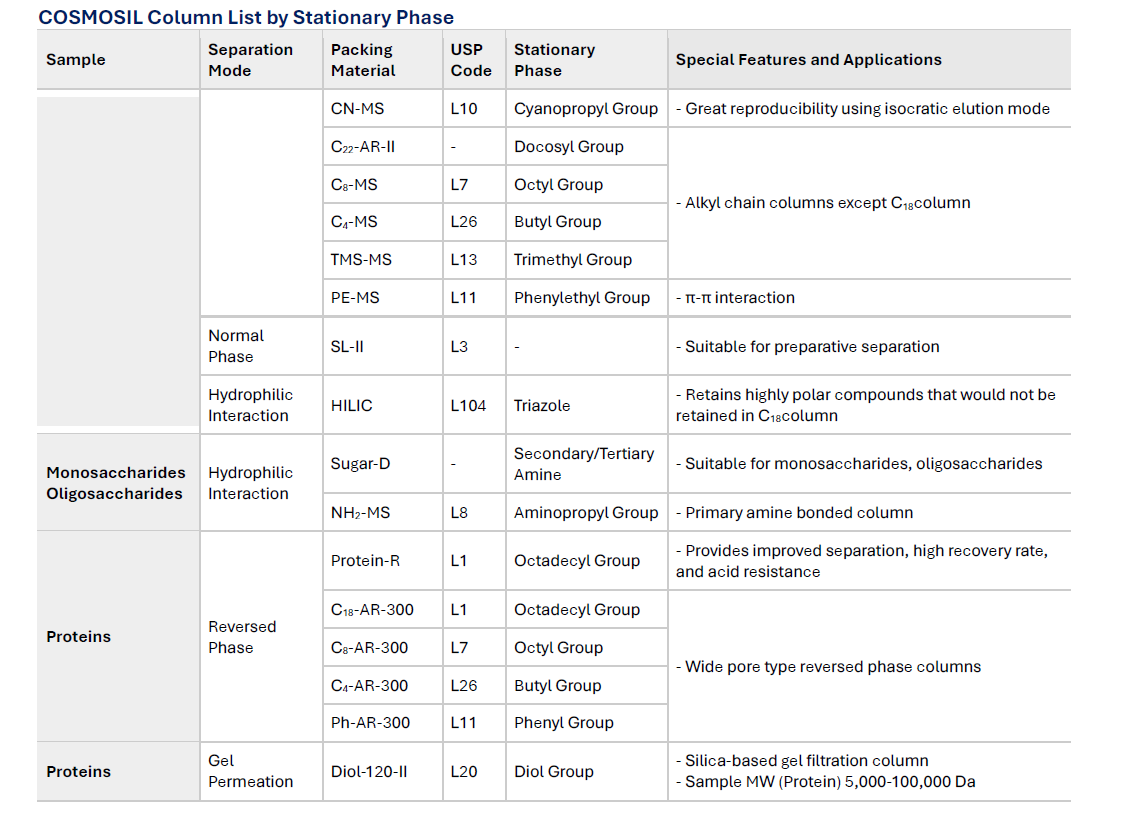
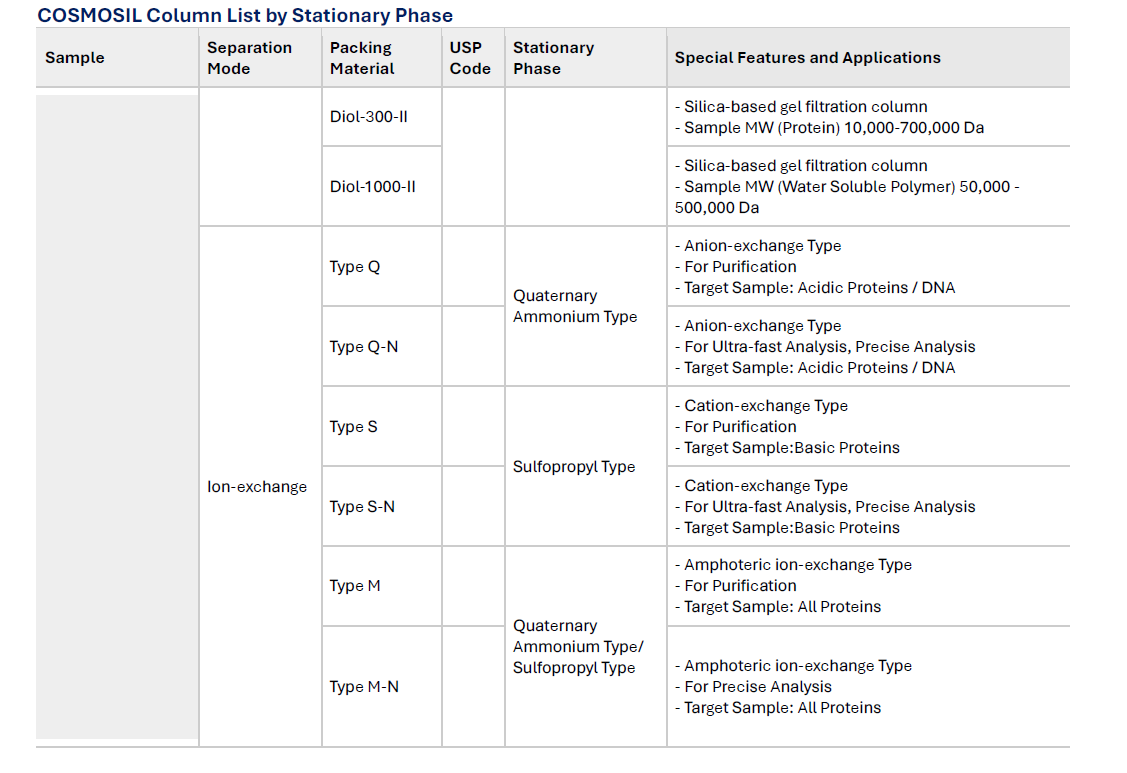
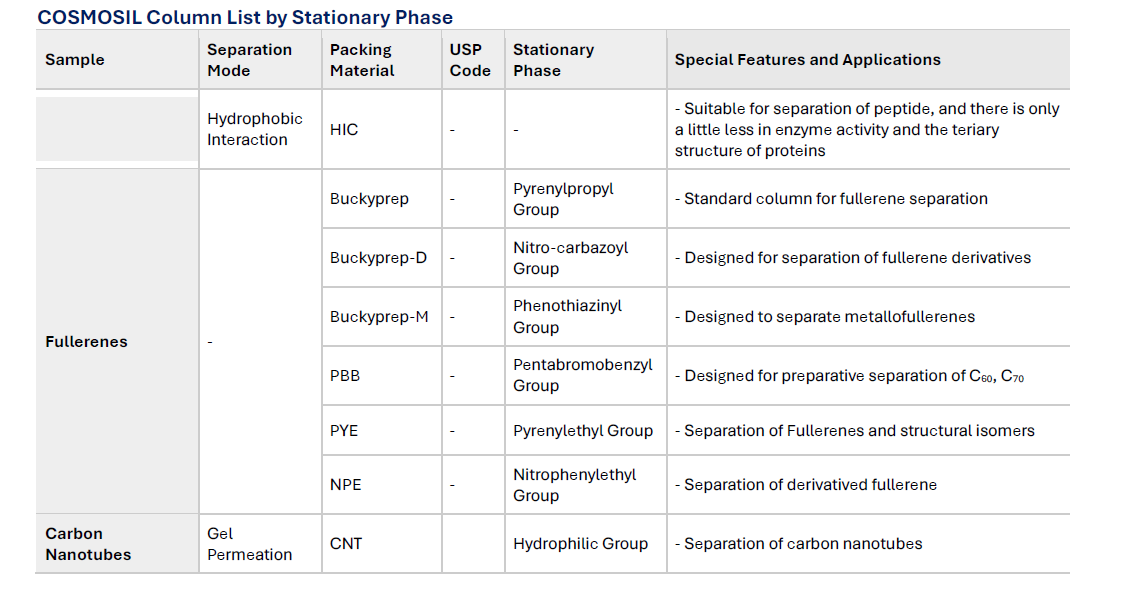
COSMOSIL Column List by Stationary Phase
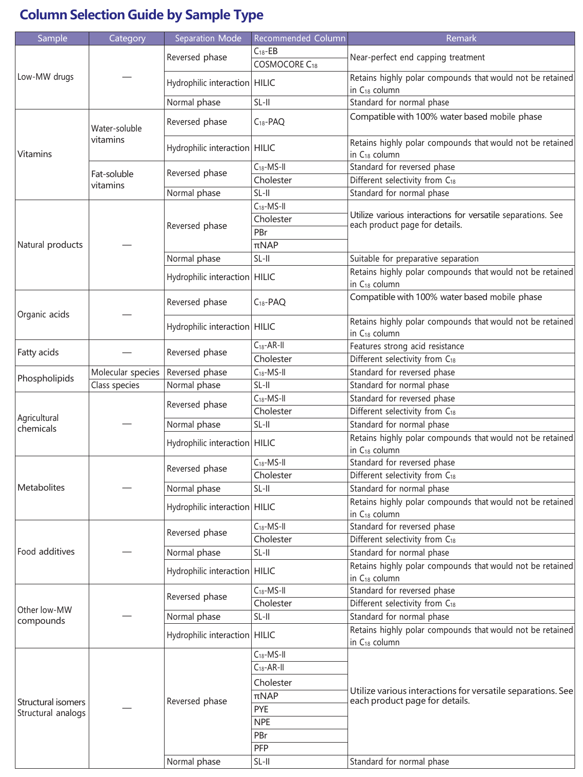
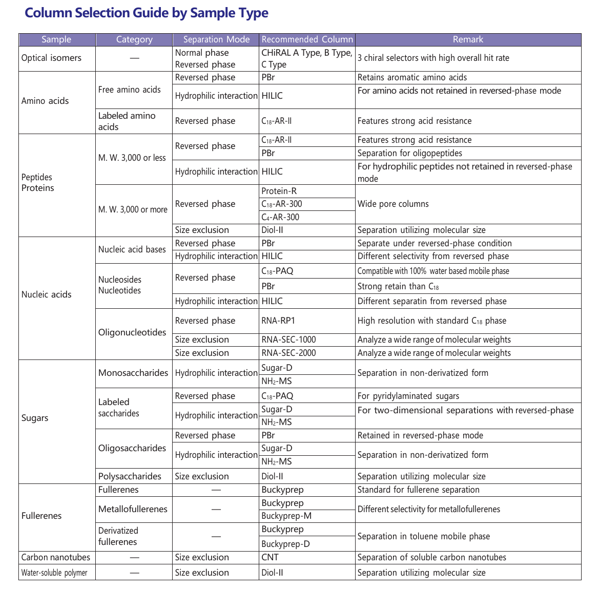
Column Selection Guide by Sample Type
Columns for USP Specifications
| USP Code | Description | Recommended Columns |
|---|---|---|
| L1 | Octadecyl silane chemically bonded to porous silica or ceramic micro-particles, 1.7 to 10 m in diameter, or a monolithic rod | COSMOSIL 3C18EB COSMOSIL C18-MS-II COSMOSIL C18-AR-II COSMOSIL C18-PAQ COSMOSIL C18-AR-300 COSMOSIL Protein-R |
| L3 | Porous silica particles, 5 to 10 µm in diameter | COSMOSIL SL-II |
| L7 | Octylsilane chemically bonded to totally porous silica particles, 1.7 to 10 µm in diameter | COSMOSIL C8-MS COSMOSIL C8-AR-300 |
| L8 | An essentially monomolecular layer of aminopropylsilane chemically bonded to totally porous silica gel support, 1.5 to 10 µm in diameter, or a monolithic silica rod. | COSMOSIL NH2-MS |
| L10 | Nitrile groups chemically bonded to porous silica particles, 3 to 10 µm in diameter | COSMOSIL CN-MS |
| L11 | Phenyl groups chemically bonded to porous silica particles, 1.7 to 10 µm in diameter | |
| L13 | Trimethylsilane chemically bonded to porous silica particles, 3 to 10 µm in diamete | COSMOSIL TMS-MS |
| L20 | Dihydroxypropane groups chemically bonded to porous silica particles, 5 to 10 µm in diameter | COSMOSIL Diol-120-II COSMOSIL Diol-300-II COSMOSIL Diol-1000-II |
| L26 | Butyl silane chemically bonded to totally porous silica particles, 3 to 10 µm in diameter | COSMOSIL C4-MS COSMOSIL C4-AR-300 |
| L43 | Pentafluorophenyl groups chemically bonded to silica particles by a propyl spacer, 1.5 to 10 µm in diameter | COSMOSIL PFP |
| L99 | Amylose tris-(3,5-dumethylphenylcarbamate), immobilized on porous, spherical, silica particles, 3 to 5 µm in diameter | COSMOSIL CHiRAL A |
| L101 | Cholesteryl groups chemically bonded to porous or non-porous silica or ceramic micro-particles, 1.5 to 10 µm in diameter, or a monolithic rod. | COSMOSIL Cholester COSMOCORE Cholester |
| L104 | Triazole groups chemically bonded to porous silica particles, 1.5 to 10 µm in diameter | COSMOSIL HILIC |
| L119 | Cellulose tris-(3,5-dichlorophenylcarbamate), immobilized on porous, spherical, silica particles, 3 to 5 µm in diameter | COSMOSIL CHiRAL C |
Conventional Columns versus High Performance Columns
A period of more than 40 years has passed since the first COSMOSIL 5C18 columns were developed and offered for sale. Continuous technical improvement has made many of these columns obsolete and not of the highest quality and performance available any more. However, many long-term users continue to employ these older conventional columns for routine analysis and quality control. Nevertheless, the manufacture of these older columns will eventually cease and we strongly urge customers to replace the conventional columns with their higher performance equivalents outlined in the table below. For additional information, please contact us.
| Conventional Columns (Old) | High Performance Columns (New) | |
|---|---|---|
| 5C18-AR | → | 5C18-AR-II |
| 5C18 | → | 5C18-MS-II |
| 5C18-MS | → | 5C18-MS-II |
| 5C18-P | → | 5C18-PAQ |
| 5NH2 | → | 5NH2-MS |
>> Click here for ordering information of Conventional Columns (old).
COSMOSIL Application Search
COSMOSIL Application, which includes more than 7,700 applications using COSMOSIL/COSMOCORE columns, is now usable by clicking the link below which is in Nacalai Tesque, Inc. The application is searchable by sample category, sample name, CAS No., column name and particle size